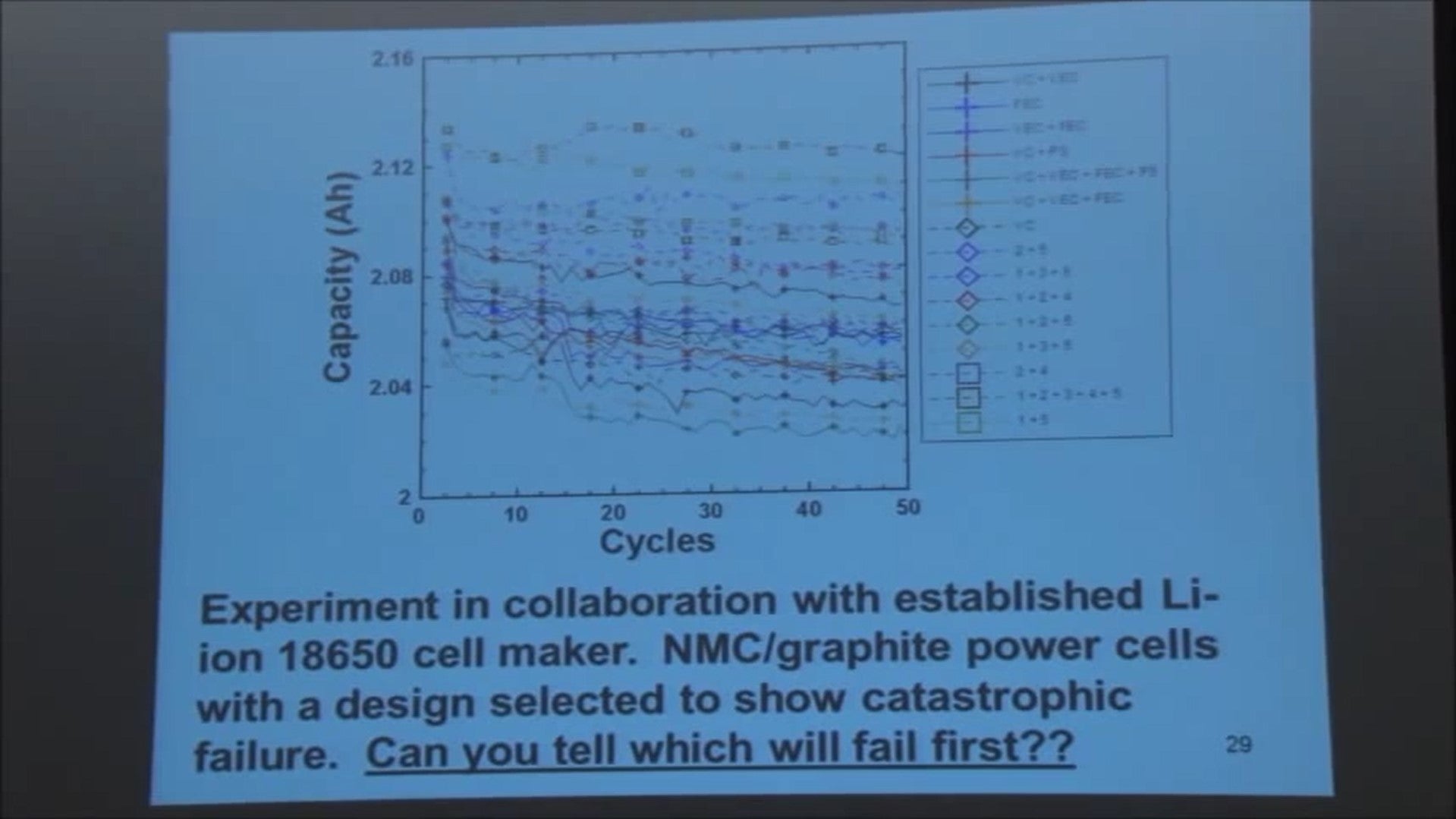
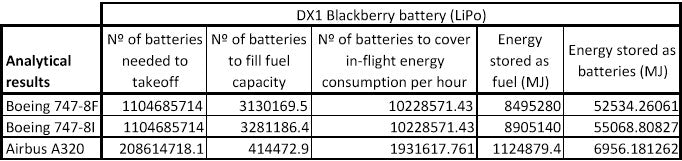
Exactly one of these LG HG2 18650 batteries is fake. Which one do you think is most suspicious?
- Can you tell which one is fake just by looking at this picture?
- Does anything in this photo immediately raise red flags?
- Are there any simple tests you can do yourself without special tools?
For the rest of this blog post, these cells will be displayed in this order, labeled Cell #1, Cell #2, Cell #3, and Cell #4.
Please take everything from this point on with a grain of salt. It is far from an absolute guide. Our observations are for a small selection of HG2 cells. Furthermore, with every new production batch that comes to market - things may change including markings, PVC, grooves, and so forth.
Overview
Ever since we posted a review of the LG HG2 battery, we have been asked the same question over and over- "how can I tell if my HG2 is fake?"
It is a good question to ask indeed, as we have identified a seemingly endless number of counterfeit HG2 cells on the market. It can also sometimes be very difficult to tell the difference, even for experienced purchasers.
How we obtained the cells for this article
Battery Bro is an American team in Asia. I recently stayed in Dongguan, China where I had the privilege of ordering things with Taobao. For those of you who don't know, Taobao is the largest e-commerce website in China, and well-known for the number of fake things.
Chinese 18650 wholesalers usually have two sources of revenue - their domestic sales (where they host a store on Taobao) and their international ones (selling via Alibaba, cold-emailing, and so forth). Ordering from a Taobao shop is an easy and anonymous way to verify supplier stock.
What counterfeiters will do to better cover their tracks is to make sure sample orders for buyers always contain authentic cells. And when you start ordering larger quantities they will start intermixing fake cells with real ones. This makes it quite difficult to identify fakes, and impossible for most overseas buyers.
On Taobao however, things are different. Suppliers will gladly sell fake cells with their arms wide open. Domestically there are far fewer repercussions and chances to be caught than when sending them through international customs.
So we anonymously purchased cells from as diverse a seller group as possible (all who are listed on Alibaba), and quickly landed fakes.
What this blog post is about
In this blog post we will take four different HG2 batteries that I think are characteristic of the overall stock on the open market. This will hopefully answer some important questions and help you identify whether yours is fake or authentic.
Remember, three of these batteries are genuine, and only one is a fake. If you want to know which one is fake right away, just scroll to the end where we provide a summary of our findings.
Visual inspection & other measurements

When I lay them down I can see that general characteristics of all the cells are the same. I am mainly looking at the positive terminal and seeing if there are any significant differences at a glance.
Underneath the positive terminal is the vent, and sometimes there are markings here which can help identify the cell. Markings near the vent can range from grooves on a certain number of sides to markings along the actual vent.
Unfortunately in this case I did not find any marks under the top-cap which will help us quickly identify fake HG2 batteries.

Here is a close-up of the top caps (positive terminals) of the cell.
All HG2 cells have four points of connection on their top cap.
Note that all their washers or insulator rings all have a textured, rather than a glossy finish.
You can run your fingernails and softly scratch them, or hold them up to a bright light and try to catch a gleam to characterize your own cell.
The washer is textured

Here is a close-up of the texture on cell number 3. Notice how there is no shine and the texture is not very smooth.

Here is a picture of a confirmed fake HG2 cell we received from a vape shop owner. They emailed us, asking us to confirm whether they had received fakes, as these cells were showing a weak battery error in some mods, while other lower capacity cells were not.
The washer on this HG2 is shiny with a glossy finish. None of the genuine HG2 cells we have recently tested have a shiny washer so this should be a tell-tale red flag.
Looking for wear and tear and other markings on the bottom terminal

This is an image of the negative terminals of the cells.
Please note that the first two cells have already been charged and discharged at the point this picture was taken. This is so you can clearly see the difference that wear and tear has on new cells.
The steel tube scratches easily, and if there are an unusual number or an "organic" arrangement (ones were are clearly not created by robotic machinery at LG Chem - usually circular and limited to the center) of scratches, then you may be suspicious of a Grade B or Grade C (refurbished) cell.
Comparing top-cap grooves

If we move back to the top caps, we can observe the grooves which are formed by the connection between the positive and negative terminals. We can also see any other etching or grooves created by the machinery which assembles the steel tubes and cells.
In this image, I can clearly see a difference in Cell #2, which has grooves which extend further than the others. Please take a moment to observe the difference for yourself.
Measuring height of 18650 batteries with digital calipers

If one cell has a different length of grooves, perhaps the overall length is different. So let's measure all four cells and quickly check.
Cell #1 measures in at 65.4 mm.

Cell #2 has a height of 65.2 mm.

Cell #3 has also has a height of 65.2 mm.

And the last cell measures 65.3 mm. Looking at the HG2 spec sheet, the cells should measure as follows,
Official specification:
- Height : 65.0 ± 0.2 mm ( Max. 65.2 mm )
That means Cells #1 and #4 do not pass.* Although, the amount is very small (.1mm and .2mm respectively) - I believe it is due to measurement error on my own part, as all the cells seem to be measuring around .2mm too long.
*Thank you /u/SCalderwood for this correction
Now, let's also measure the cell's diameter and weight.
HG2 diameter

As you can see, all batteries have exactly the same diameter - 18.3mm
Compared to official specifications:
- Diameter : 18.3 + 0.2/-0.3 mm ( Max. 18.5 mm )
All the cells are the correct diameter.
HG2 weight

The weight measurements are as follows:
- 45.05 g
- 44.55 g
- 44.88 g
- 44.75 g
Official HG2 specifications:
- Weight Max. 47.0 g
Since only a maximum weight is given by LG Chem, and none of the cells have serious variation we can say the weight tests pass for all cells.
Looking closely at the PVC

Here is that image from the beginning of this article. If you noticed the color of the second cell seemed a little off, you are not alone. The color does indeed seem to be different.
If the PVC is fake, where is it coming from?
The color of the battery comes from something called a PVC heat-shrink tube and the mostly neutral metal steel behind it.
There are a few different ways counterfeiters can make fake LG tubes, so let me quickly describe them. Creating high-quality PVC requires expensive tools. A PVC injection mould machine (just a small one) will cost upwards of $15,000, not to include the expertise, parts, and other materials required to run it.
It is for this reason I speculate that high-quality brown-colored PVC tubes are not coming from any low level 18650 battery traders. Brown PVC tubes that mimic those that LG produce are probably coming from two sources:
- 1.) A factory that is commissioned to LG to produce their PVC, also produces excess and sells to counterfeiters behind closed doors.
- 2.) A PVC company that either: A.) Sells counterfeiters blank tubes of a particular desired color, and the counterfeiters print the text themselves, or, B.) Sells counterfeiters tubes with the trademark infringing text already on them.
In all cases, the 18650 counterfeiters are purchasing the actual PVC rather than producing it themselves.

In scenario 2A, a counterfeiter could purchase a cheap rotogravure machine and print any text they want. But the quality might be low, and the other two options are more plausible for high-quality fakes.
Now, let's take a closer look at the color difference between the cells.

Try to notice the difference in color that Cell #2 exhibits in this shot.
The light-sources used in this photograph, along with the shadows and reflections from an uneven environment can all obscure the true colors of the cells. For this reason, you should take a look at the next shot, flip back and forth, and try to notice the difference.

Cell #2 seems more purple, or more red, than the other two doesn't it?

In this black light test, no abnormalities were detected.
For this test I have cut some PVC off of each cell and shined a very bright colorless light through each one. This is so we can now compare the color difference more accurately on a computer.
Please note, they look more red than brown now because of the amount of light penetrating the PVC (it's like when you add milk to your hot chocolate, the brown gets lighter). In this case, the brown hue on the PVC is created from mostly dark red tones, and the red tones have been brought out more with additional light.

On a black background, it is easier to pick up some of the differences in tone.

After digital expansion, you may compare them to each other, rather than the black or white backgrounds.
Cell PVC #2 is the outlier and seems to have the most distinct and vibrant red color. The other three PVCs all seem to have more blue and green pigment in them.

Using a black and white infrared filter, we can clearly now see that the PVC on Cell #2:
- Has the least green/blue pigments
- Has the most red pigment
So basically, Cell #2 (the suspected fake) has more red in it. This increased red hue, mixed with the other colors, makes the cell appear more purple in real life.
Unwrapping the HG2

I like to use a needle, as it lets me precisely remove the PVC without causing damage to the battery. A knife or larger instrument might easily scratch the cell.
I recommend you do not do this at home. If you do want to unwrap the cell yourself, you should choose a non-metal tool, like a ceramic knife or something else that will not conduct electricity.
You should also have a new PVC to replace it with immediately to prevent short-circuiting in your device, trash bin, or storage container.
Tip: When unwrapping a cell, always start at the negative terminal and work your way upwards. Avoid using any tools that conduct electricity at the positive pole of the battery. This goes for both unprotected cells, and when removing the PCB from a protected cell.

After the cells are unwrapped, there are clearly some differences:
Top-cap grooves:
- We can now clearly see why the top-cap grooves on Cell #2 were longer. The gap where it is fixed to the negative terminal is larger, as well the ring of smoother metal sitting just under the gap appears to be larger.
- This ring of smooth metal under the top-cap gap is created by robotic machinery and the comparative difference between Cell #2 and the other cells is indicative enough in this case to cause suspicion.
- I am not providing measurements of these grooves because they vary so much, and can be very misleading. I have seen genuine HG2 cells with the larger grooves as well. However in this case, the comparative difference is what is interesting.
Quality control and identification markings
Cell #1 has absolutely no markings whatsoever. This should normally raise a red-flag, however in this case it can be explained, and I will do so in a moment.
Cell #2 has the most markings. These markings present on all sides of the steel.
Cells #3 and #4 both have aesthetically identical markings, only slightly varying in alphanumeric value. Over the last several weeks we have seen this style of markings on genuine cells. Also, consider that the markings only vary slightly (I1022A vs. I11214) This adds confidence that these two cells are from the same production batch.
Unfortunately I must stress, that markings on steel can also be faked quite easily.
Why do I say this? Take a look at the following image:

Remember the vape shop who sent us photographs of their fake HG2 with the shiny washer / insulator ring? Here is another picture they sent us, this time of the markings.
The markings here are quite similar to the markings on Cells #3 and #4.
The difference then we can focus on, would not be the alphanumeric values of markings themselves but the quality of the markings. The marking on the cell shown above is faded, washed out, and unclear compared to those in Cells #3 and #4.
This is not an absolute indicator, but can be a form of corroborative evidence you may use.
There are two ways for markings to be faded, washed out, and unclear.
- The cells are genuine, but the machinery needs tuning, or the ink is simply running low (just like in the InkJet printer at home, this happens when printing everywhere including in a large factory).
- The cells are fake, and the equipment used to print are lower quality, hence the faded appearance. The machinery counterfeiters use are almost always lower quality than what LG Chem uses.
Why does Cell #1 have no markings?
Markings on the steel are an excellent and-time tested way to add evidence to a cell's authenticity. That is because, it is always easier for a counterfeiter to purchase a blank steel tube and not print on them. Or to use a different battery. In some cases the counterfeiter will file off any markings, which will appear rough and slightly depressed from the original surface.
If you find any indication that markings have been filed off the surface of your 18650 battery, this is strong evidence of a counterfeit.
However, please remember that markings in most cases are to be taken with a grain of salt. Markings can not alone determine whether a cell is fake or not (as they can be faked quite easily themselves). They should only be used as a weak form of corroborating evidence.
HG2 Timeline
There have been, for several months, a large number of HG2 batteries with no markings. They started appearing at a time when the HG2 was very scarce. The great scarcity of the HG2 was between the end of chinese new year (around February 15th) to late March 2016, and was caused by a number of factors.
During this period, nearly every Chinese supplier had run out, causing a shortage in other most other countries like the US.
However, around the same time, a large batch of HG2 batteries had been sent to Japan for a certain 18650 pack assembler, which ordered in excess. These cells in Japan were sent directly from South Korea to a specific manufacturer, were specially produced, and thus for their purpose never had any identification markings printed on them.
This particular manufacturer in Japan either by luck or by savvy sold off many of their cells at a time of scarcity, and thus the market has seen tens of thousands of LG HG2 batteries without any markings.
These batteries are authentic, Grade A, and yet had no markings.
| February 1 | LG rumored to cease HG2 production for retooling (high percentage of rejects) |
| February 7 - 13 | Chinese New Year |
| February 15 | SK/HK Distributors cease HG2 sales |
| March 15 | Unmarked HG2s appear |
| March 25-30 | New HG2s appear |
| April 1 | New IATA air shipping regulations take effect; air shipping of HG2s halts |
| April 10-20 | HG2 supply dwindles again |
But why does Cell #2 have so many different markings?
In my opinion, it is because it is a different cell. And I say that for a simple reason, and it's that we have seen these markings before, on a different cell manufactured by LG.
But I will return to this question a little later. First, let's see if there are any other abnormalities under the wrapping.

The top-caps all look the same, albeit the ring where the washer is meant to sit on Cell #2 is a bit cleaner and lighter than the other three cells.
Also, that the gap and shiny ring where the positive terminal and negative terminal meet is now confirmed to be notably larger.
I'm going to start calling Cell #2 the fake from this point on, although it is still unconfirmed I want to let you clearly know where the suspicion is. This is because we have identified three types of weak corroborating evidence:
- Different coloring on the PVC
- Different groove, top-cap spacing, and metal appearance under where the washer would normally lay
- Different markings on steel tube

Here you can observe the difference between the two 18650 batteries with enhanced sharpness.

Here is another close-up shot with enhanced sharpness.

Compare with Cell #3.

And remember the battery picture from the vape shop we got? The one with the shiny (not textured) washer? This is a photograph of the top-cap of that cell.
If we look closely at the metal where the washer would normally lay, it clearly looks cleaner. The genuine cells we are looking at have a much browner appearance here, while the fake ones have a more of a metallic steel color.
Take a moment to scroll back up and look at the more brownish color that Cell #3 exhibits here.

Another shot of the markings on Cell #3.

And the opposite end of HG2 Cell #3. Note the markings are scarce on this cell, and not seen all the way around.
What could Cell #2 possibly be if it is not an HG2?
Earlier I had said, I have seen markings such as the ones on Cell #2 before. Please take a look at the following images and judge for yourself:

On the left of this image is Cell #2, turned 360 degrees to show all of its markings.
On the right side is an unwrapped HE2 we did for a previous post. Can you notice the similarities?

Here it is, a little bit easier to compare the markings.

What is some other evidence pointing the fake HG2 as actually an HE2 or HE4?
So could Cell #2 be an HE2 or HE4, rewrapped as an HG2? Would there be motivation for counterfeiters to do so?
Motivation for using an HE2
There are two big reasons - availability and cost.
The cost difference between the LG HE2 vs the LG HG2 can be upwards to $1.00 USD, making the HE2 substantially cheaper. Since wholesale 18650 batteries are a commodity, and they are available on the open market, a price difference of up to a dollar is extremely significant to the profit margins of counterfeiters.
Furthermore the HE2 is a couple years older than the HG2, and is readily available on the market. There are no shortages for the HE2, and it is a highly liquid cell. Whereas, the HG2, even though they have become more available are still relatively scarce and seeing shortages.
The HE4 is slightly more expensive, and less available than the HE2.
Other reasons to use the HE2 as a fake HG2
Furthermore, if you are going to fake an HG2, what is the easiest cell to use? Another LG cell is - because its top cap is going to be identical or nearly identical (with four positive terminal connection points).
If you were going to use a Samsung, or Panasonic cell the top-cap would be a dead giveaway as they use a different number of connection points and styles.
The last point in the favor of the HE2-as-a-fake-HG2 hypothesis is that they are both 20A cells. The counterfeiter may not want exploding batteries (end-users discharging too much current) as this would draw unwanted attention., and thus choosing one with a similar maximum continuous discharge rating is favorable. The loss to the consumer, is one of 500mAh. (The HG2 is rated at 3000mAh, while the HE2 is rated at 2500mAh - both are rated at 20A max. discharge current).
It is for these reasons, that by visual inspection alone I can say with some degree of confidence that Cell #2 is probably fake, and finally that it may be a rewrapped HE2.
However, we can't be certain until we do some actual testing. So let's get to it!
Testing the LG HG2 cells (and possible LG HE2)
The first thing we must do is rewrap all of the cells before testing them. Again, this is to prevent short-circuiting and should always be done. Unwrapped cells are dangerous and easy to short-circuit. We use a heat-gun, but a blow-dryer works well too.

We also have to spot-weld some tabs on the newly wrapped cells so we can attach alligator clips to them.

We are using a RePower battery charge and discharge station (AC 220 / 60V / 20amp) for these tests because it will accurately show us a discharge of up to 20 amps - which the HG2 is rated for.
A 20 amp discharge should quickly be able to tell us which of the cells is counterfeit.
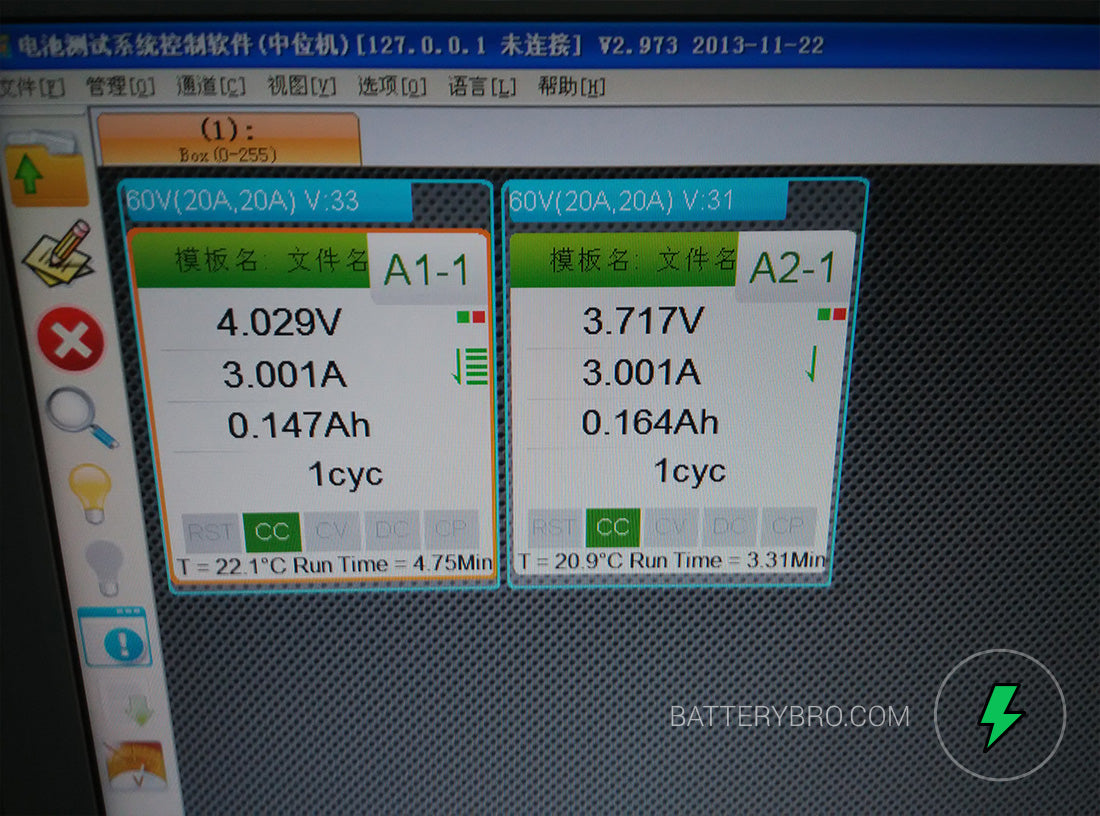
Here we go! Charging up the first two cells.
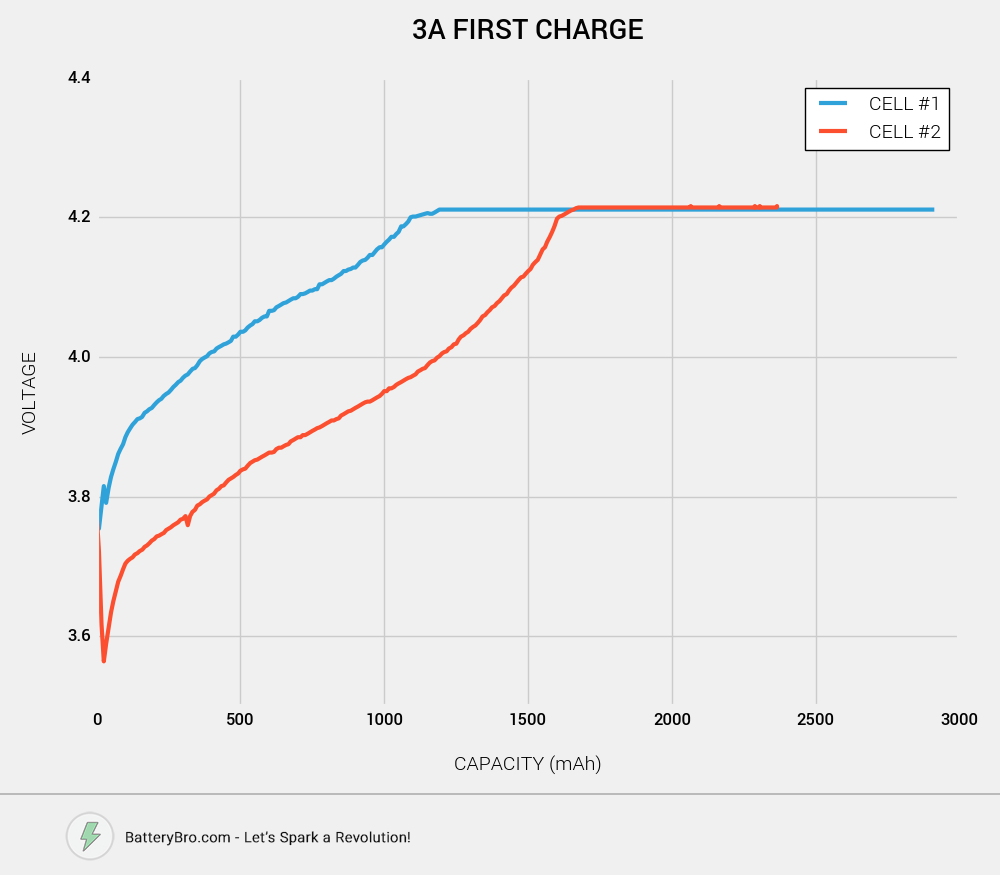
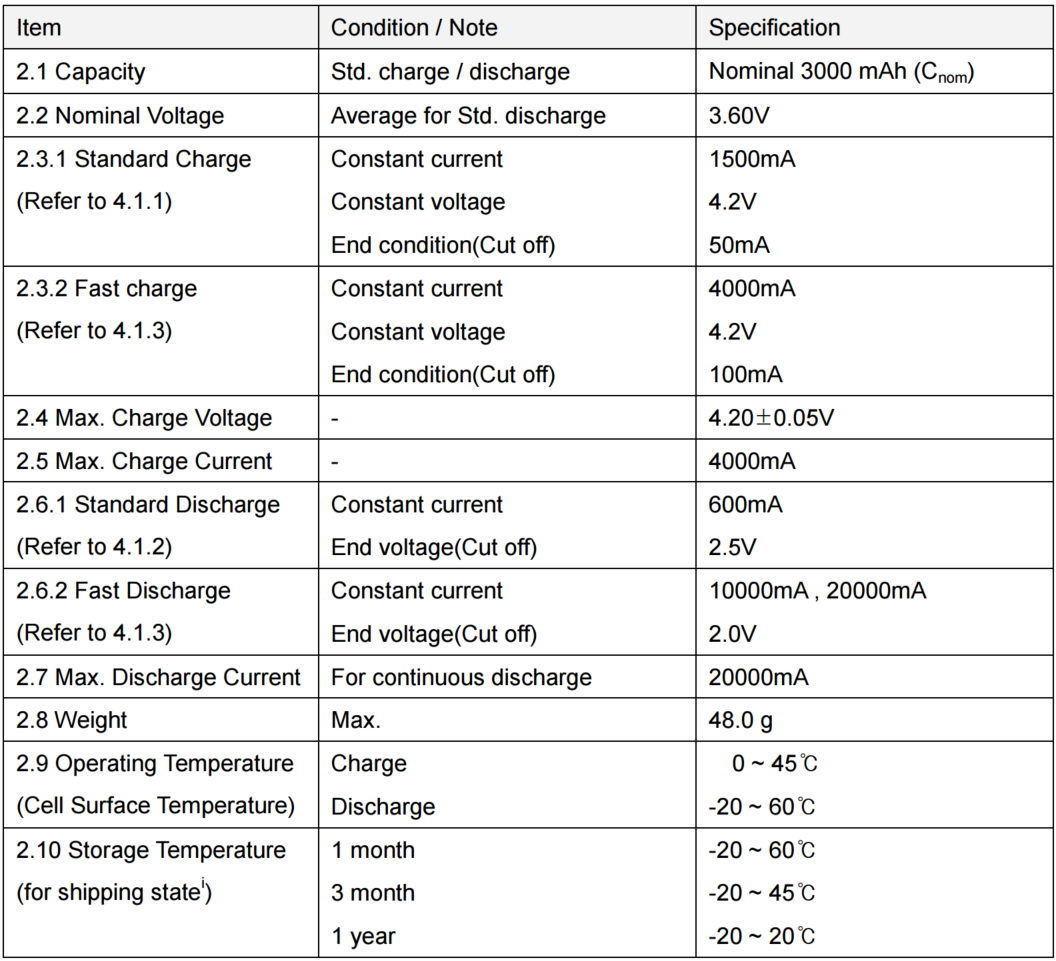
First, we had to charge the cells fully. Both cells were charged at 3A, which is very fast for charging. There are two important differences to note. First, Cell #1’s voltage rises MUCH more quickly than Cell #2. Second, Cell #2 has about 500mAh less to charge than Cell #1. This suggests that Cell #2 has less overall capacity than Cell #1.
However, this could be due to different starting capacities for both cells. Perhaps one cell wasn’t discharged as fully as the other? Let’s move on to our first discharge test at 10A.
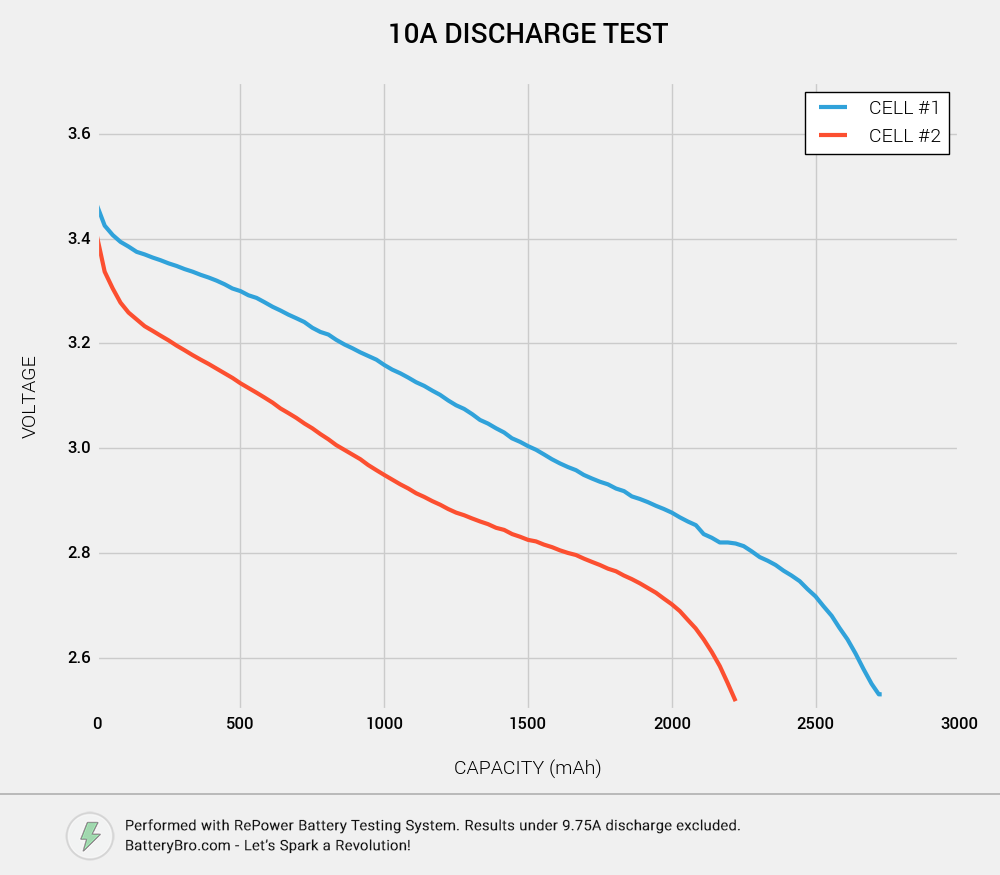
Aha! Cell #2 really does have less capacity than Cell #1! Cell #1 appears to have about 2700mAh of capacity, while Cell #2 has about 2200mAh. A 500 mAh difference… suspicious!
But if Cell #1 is an HG2, shouldn’t it discharge to 3000mAh? There are a few reasons you get less than 3000mAh here:
- When you discharge cells at high amperages, you get less overall capacity.
- This graph only shows data where the amperage is greater than 9.75A. At the end of any discharge test, the amperage tapers off. We want to see how much capacity you get AT HIGH DRAIN, so we exclude the data under 10A (as is standard practice).
Another interesting tidbit is Cell #2’s enormous voltage sag. As soon as the discharge test begins, Cell #2’s voltage sinks immediately relative to Cell #1. Not only do you get less capacity - you get far less voltage throughout the discharge cycle.
For Cell #1, this test took about 16 minutes. Cell #2 took 13 minutes to fully discharge.
(For high-level battery geeks out there: the reason the charts begin at about 3.4 volts is that our testing equipment doesn’t sample quickly enough to capture the high-voltage discharge that occur immediately after testing begins.)
But voltage is only part of the story. As we all know, excessive heat is bad for 18650s. It reduces their performance and cycle life. How do the two cells stack up in terms of temperature?
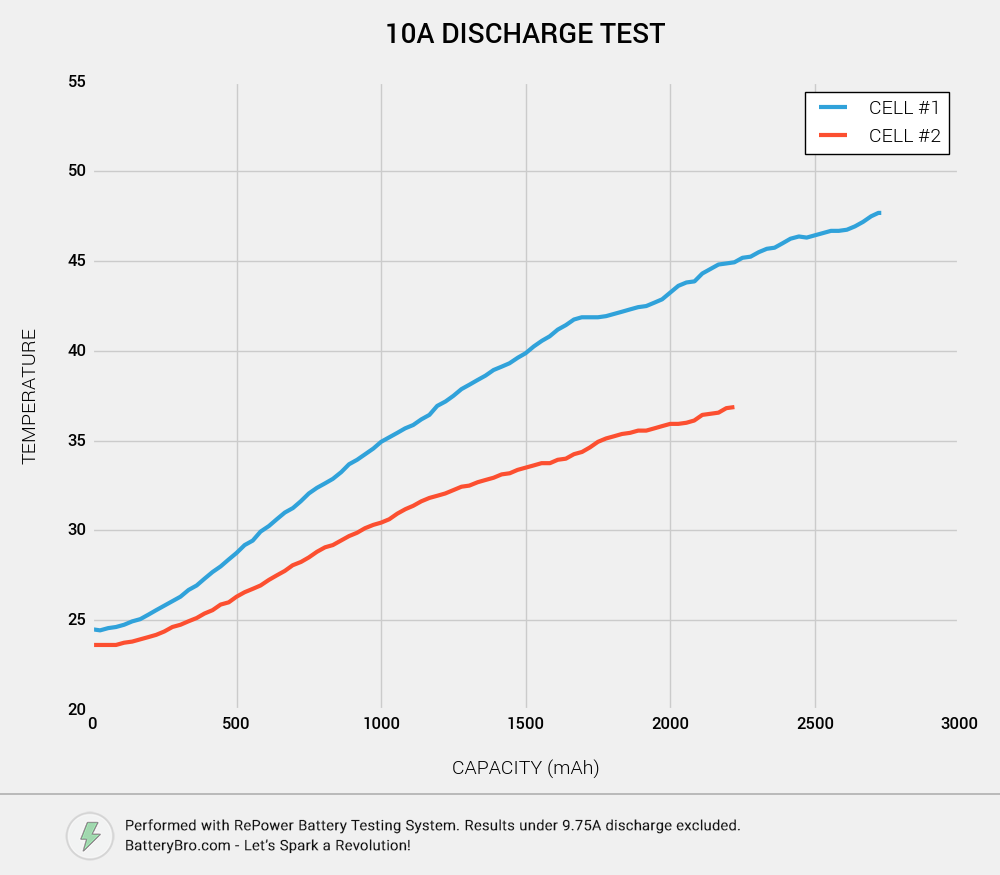
This chart shows how the cells heated up throughout their 10A discharge cycle. Cell #1 has an early, if small lead, which quickly grows into an 8 degrees Celsius temperature difference. Cell #2 doesn’t heat up nearly as much. Does that make it a better cell? The most likely explanation: Cell #2 was running at a lower voltage, and for less time.

This chart is a little more complicated. On the X axis is the cell voltage at any time during the discharge cycle; on the Y axis is the cell’s temperature. We can see that even at the same voltage, Cell #2 has a much lower temperature. This is probably due to the fact that it spends much less time at low voltages and doesn’t have as long of a run-time.
Now that we’ve seen how the two cells perform - very differently - let’s move on to the 20A discharge tests. This is the king of high-drain battery tests: the only true way to see how high-drain cells stack up.
We introduced an older HG2 (Cell #3) for this test, which has been through about 30 discharge cycles, to see whether Cell #2 might just be an old HG2.
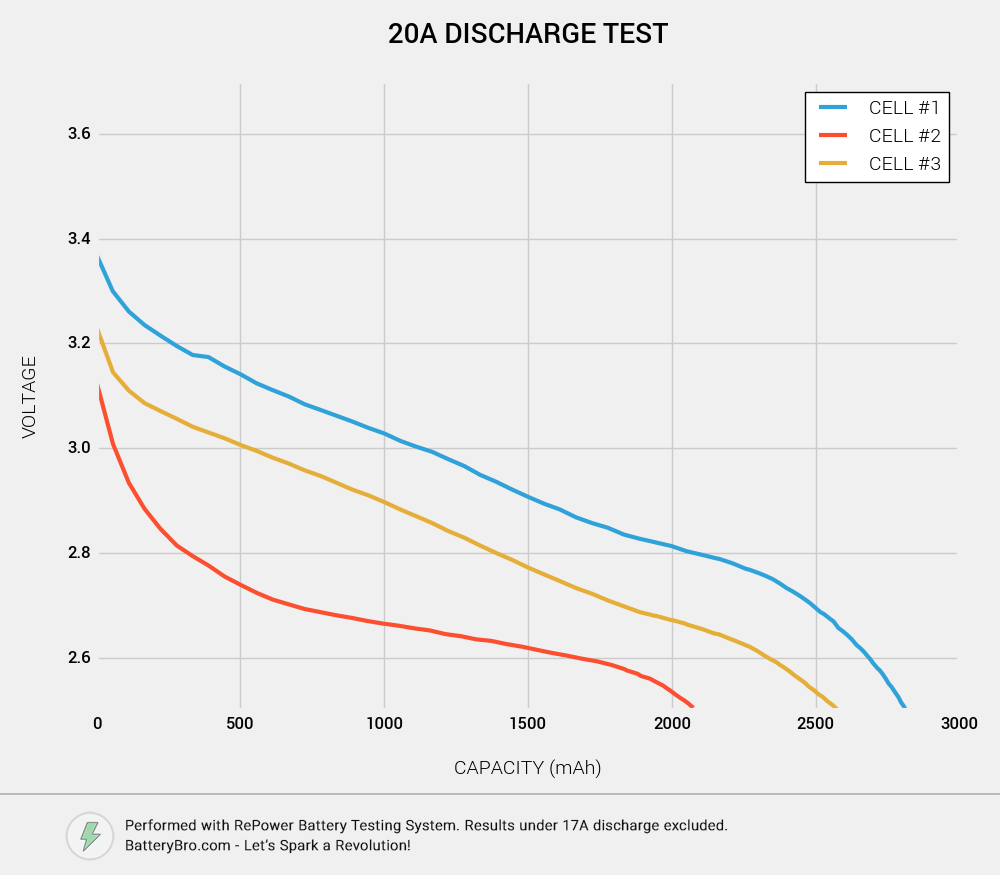
Now it’s becoming clear that Cell #2 is not an HG2. Both Cell #1 and Cell #3 show the right amount of capacity, a bit above 2500mAh: this is what you would expect from a 3000mAh nominal cell at 20A discharge. However, Cell #2 only ends up with about 2050mAh. This is consistent with a 2500mAh or 2600mAh rated cell discharging at 20A.
One thing to note is that you would normally expect a cell to have less capacity at 20A than at 10A. This was true for Cell #2, but Cell #1 showed the opposite effect. This may be caused by the fact that this was one of Cell #1’s first discharges; sometimes 18650s need a few discharge cycles before they perform at their full potential. This may also be because much more time elapsed between the two discharge tests for Cell #1 than for Cell #2. We will follow up on this with our own testing.
Another thing to note: Cell #2’s voltage sag is MUCH worse than in the 10A discharge test. Much, much worse. While Cell #1 and Cell #3 had basically the same shape in their discharge curve, and although Cell #3 underperformed voltage-wise, Cell #2 shows a much earlier, steeper voltage sag. This is the mark of a very poorly performing high-drain 18650.
Now let’s look at how the temperature charts stack up.

Once again, we see the same pattern of Cell #1 heating up more than Cell #2. However, this time the temperature difference is much smaller.
(The difference between starting temperatures is easily explainable. Both Cell #1 and Cell #2 were charged just prior to being discharged. Since Cell #1 was charging for longer - it has more capacity, after all - it started just a bit hotter than Cell #2. Cell #3 had been fully charged long before its discharge test.)
All three cells reached temperatures that would degrade performance, over the long term. But this is to be expected for a continuous discharge at 20A. Battery pack users, beware: just because a cell is rated for 20A doesn’t mean you can push it that hard throughout its discharge cycle! It will have a hugely detrimental effect on battery life.
These tests lead us to conclude that Cell #1 and Cell #2 are not the same.
Cell #1 and Cell #2 are not the same
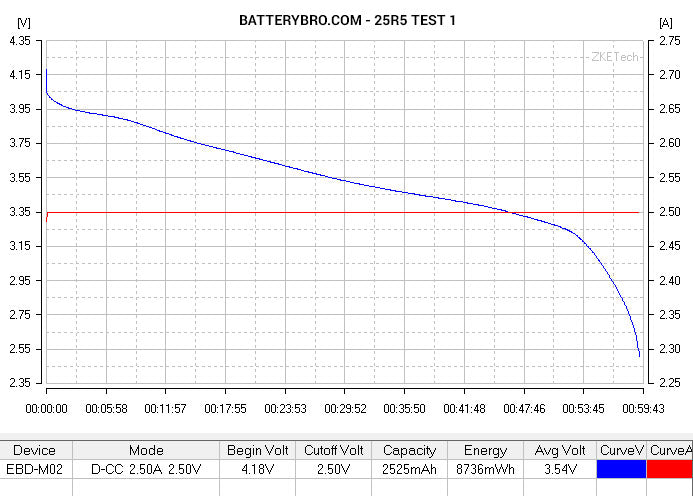
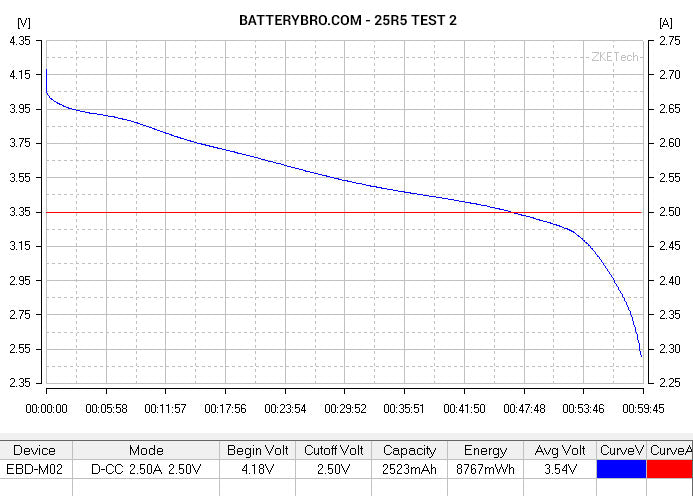
Cell #1 shows all the characteristics of a high-performing, 20A-rated, 3000mAh cell. Cell #2, not so much. Could it be a rewrapped LG HE2? Perhaps a Samsung 25R5 (unlikely given its physical characteristics)? Let’s see how it compares to data from some of our previous tests.
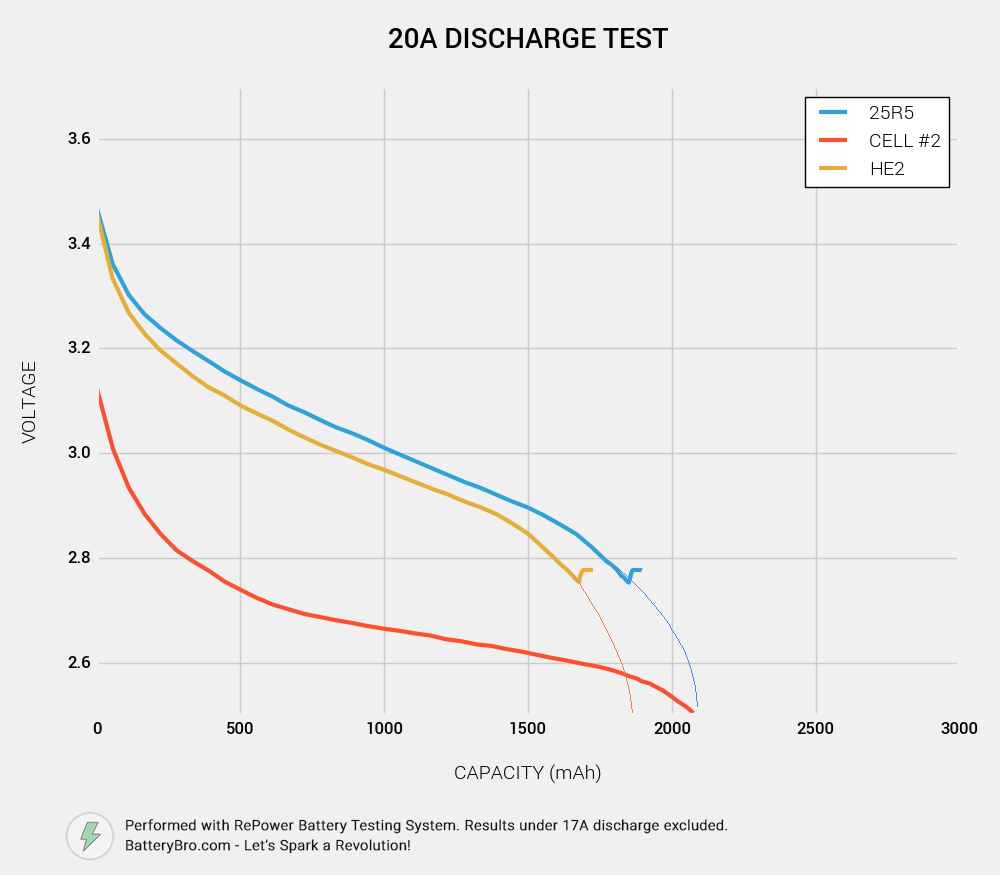
Okay, so you probably notice that the 25R5 and HE2 end much earlier in their voltage curve than Cell #2. We used slightly different parameters to run those tests - the cutoff voltage was 2.75V rather than 2.5V. But by tracing their curves down, you can see that they end at roughly the same ending capacity:
Conclusion: Cell #2 is not a 25R5 or HE2, although it has roughly 2500mAh capacity.
The incredible voltage sag suggests that Cell #2 is not rated for 20A discharge. Although it did not heat up to dangerous temperatures, it did not perform as a 20A-rated 18650 should under these discharge conditions.
What’s going on here? There are two possibilities:
- Cell #2 is an old, used LG HE2 possibly salvaged.
- Cell #2 is a cheaper LG cell that is not rated for 20A discharge.
- Cell #2 is a Chinese 18650 cell. Pretty likely, given that it was purchased on Taobao in the middle of a global HG2 supply shortage! Furthermore, the most popular Chinese cells are 2500/2600mAh with a sharp voltage sag.
While there were definitely a few errors in the experimental setup here, it was sufficient to show that Cell #2 is indeed not an HG2.
Conclusion
We started our mission in 2014 to fight against fake and dangerous 18650 batteries. However, as we get better, so do counterfeiters. Therefore it is always necessary to stay one step ahead and dig deeper. As we have seen in this article, telling fake cells from genuine cells is getting more difficult - but where there is a will there is a way.

Summary
Cell #2 is a fake LG HG2.
Cell #2 is likely a Chinese-made cell.
How to investigate your own cells
I believe, based on the results of this article, that currently the easiest test to see whether your HG2 is real or fake is to ask the following questions:
- Look at the color, does it appear as an earthy brown, or does it have a slight purple or reddish tint? A purplish color might indicate a fake.
- Is the washer / insulator ring shiny or matte / textured? A shiny washer might indicate a fake.
- Is the metal under where the washer would normally lay a dirty brownish color or a more clean steel metallic color? If it is more metallic in color, this might indicate a fake.
- Look at the grooves under the top-cap, do they appear small and thin, or slightly larger? Compare them with the photographs in this article. A larger groove and especially a large gap between the positive and negative terminal might indicate a fake.
- What markings does your cell have? If it is blank, or is a simple line of text such as 'I0122A', or does it have many markings all over its body? Look at the markings, and see if they match that of an HE2 or HE4. Look at the quality of the markings, are they faded and smeared? If the markings on the steel are faded, it might indicate a fake.
Finally, can you characterize your cell's performance?
- If you purchase a charger with an mAh read-out, you are able to check your charge capacity and quickly verify a cell. For example, some HG2 cells we tested only charged up to 2500mAh. This is a very quick and easy way to verify your 18650's authenticity.
- How does the cell compare to other cells you own in terms of capacity (run-time) and voltage sag?
Add up as many of these indicators as you can, the more you can check-off, the more likely you have a fake HG2 - with actual performance always being the strongest form of evidence.

A charger like the VC2 can quickly tell you if a cell does not have the right capacity.
Thanks for reading!
Feel free to comment with your own results
If you find our results controversial, have questions, or have spotted something we haven't please let us know in the comments. If you have inspected your own HG2 cells, please post your findings below to help others which come across this information.
Image usage rights
If you would like to use any of the images presented here on your own blog, website, or social media please feel free. All we ask is that you provide a link back to our website.
A special thanks to Daniel Tok from InnoVape who let us use his images of a fake HG2 for this blog post.
If you are interested in getting a quote from Battery Bro for guaranteed Grade A, authentic, HG2 batteries for your business please follow this link.
]]>